As long as pharmaceutical drugs have existed, so have side effects. Some of these are mild and considered worth the risk. Others are far more dangerous and even lethal. The worst part? The manufacturers often KNEW that the drugs were dangerous and sold them anyway.
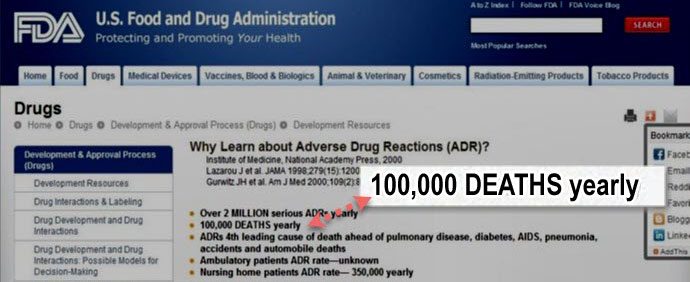
And the FDA is well aware of the deaths that some of these “killer drugs” are causing. They even admit, on their own website, that over 100,000 Americans die annually from ADR (“Adverse Drug Reactions”). Check it out!
If you do the math, that’s an average of almost 274 people every day that die from FDA “approved” drugs! Let that sink in!
Zantac
People who take Zantac or similar drugs to combat heartburn may be unknowingly giving themselves cancer. In 2019, the FDA announced that it had detected a cancer-causing contaminant called N-nitrosodimethylamine, or NDMA, in ranitidine heartburn medications.
But the FDA fell short of ordering a recall, and it wasn’t until a week later that distributors began to pull the drugs off the shelves. In response to the discovery, Sanofi spokeswoman Ashleigh Koss said,
“Sanofi takes patient safety seriously, and we are committed to working with the F.D.A.”
She went on to say that Zantac “has been around for over a decade and meets all the specified safety requirements for use in the O.T.C. market.”
At the time, Koss stated that Sanofi had no plans to recall the drug. But as evidence and consumer outrage grew over the following week, pharmaceutical company Novartis decided to stop the distribution of its generic Zantac drugs in all markets. At the time of publishing, Sanofi and the FDA had still failed to issue a recall.
This is not the first time that NDMA has been found in popular drugs. In 2018, blood pressure medication valsartan, sold under the brand name Diovan, was also found to be contaminated. In that case, the FDA issued a voluntary recall of the drug, citing the risk of developing cancer.
Millions of people take ranitidine to relieve symptoms of heartburn and other gastrointestinal issues. But the potentially life-threatening drugs can still be found in pharmacies across the United States. In Canada, health officials have requested a stop to all distribution of the drug, making it the only country in which Sanofi has ceased distribution.
NDMA is an industrial byproduct that can often be found in cured meats like bacon. The FDA says that it is “reasonably safe” to ingest up to one microgram a day. But safety testing has found levels significantly higher. In the 2018 valsartan recall, the FDA found up to 17 micrograms of NDMA per dose. Valisure, a pharmacy that tests all drugs it distributes, found that Zantac had NDMA levels reaching 3,000 micrograms.
Valisure petitioned the FDA to recall all forms of ranitidine, though the agency has yet to take any such measures. They claim that NDMA may be “inherent” in the ranitidine molecule and have urged regulators to recall the drug until its safety can be guaranteed. But the FDA, as usual, seems apathetic about consumer safety.
FDA spokesman Jeremy Kahn said in a statement,
“The FDA will take appropriate measures based on the results of the ongoing investigation.”
But they failed to recommend that people using the drug stop taking it. Instead, they simply suggested that alternative medicines are available.
This is the same “profits over patients” approach that we’ve seen repeatedly with our regulatory bodies. Safety testing – especially for generic drugs – is significantly lacking. Dinesh Thakur is a drug-safety advocate and whistleblower who exposed corruption and faulty quality control as an executive at Ranbaxy Laboratories. He says the FDA is not doing enough to protect consumers.
“I think this is another good example of how our regulations need to change. Things like this will never get caught, unless somebody is actually actively looking for stuff.”
Meanwhile, a lack of oversight and testing standards has resulted in pharmacy shelves filled with drugs that can literally kill you. The FDA knows that Zantac has been contaminated with a carcinogen. Sanofi knows that something has gone terribly wrong with one of their flagship drugs. But the money is too good to recall them now.
Zantac was the first drug to total $1 billion in sales. In 2018, the drug generated nearly $130 million dollars for Sanofi. But a new class-action lawsuit filed earlier this week claims that the French drug makers have known the risks all along, hiding them from regulators and consumers in order to maintain profits.
The suit claims that each 150mg tablet of Zantac contains 26,000 times the FDA-approved daily limit of NDMA. Steve Berman, the lead attorney for the case, believes that consumers have been intentionally put in harm’s way.
Millions of people in the U.S. suffer from heartburn, and for years, Zantac has been sold to the masses as a safe and easy-to-find remedy for that common ailment. We’re certain that if those millions of consumers knew that the Zantac they take contains known carcinogens, they would be rightfully outraged.”
“Sanofi knew that Zantac contains a carcinogen, yet it chose to conceal these risks to the public to line its own pockets,” the suit claims.
Had defendants disclosed that Zantac results in unsafe levels of NDMA in the human body, no person, let alone a reasonable person, would have purchased and consumed Zantac.”
But all statements from the FDA and Sanofi say basically the same thing:
“We care about consumer safety. We’re looking into it.”
Talcum Powder
Health and wellness giant Johnson & Johnson knew that their products caused cancer and chose to keep that information secret while manipulating research and regulations to protect the company. Executives allowed consumers to become ill and die, even going so far as to lie in court, in order to protect the company’s reputation and bottom line.
According to an investigation by Reuters, Johnson & Johnson knew for decades that its signature baby powder contained asbestos and kept the information from regulators and the public. Though executives at the company have denied the report, Johnson & Johnson stock fell nearly 13% over that weekend, costing more than $50 billion in market capitalization.
Talc, one of the softest minerals on earth and the primary ingredient in J&J’s baby powder, is mined from underground deposits. Asbestos is a dangerous carcinogen responsible for causing mesothelioma, lung cancer, and ovarian cancer. Asbestos is also found underground and can often be found in talc deposits, creating a risk for cross-contamination.
J&J has been sued countless times over claims that their baby powder causes cancer, but plaintiffs were often unable to obtain the internal records they needed to prove the company’s guilt. Several of these cases have gone to trial, including a case this summer in which 22 women, who blamed asbestos in the baby powder for causing ovarian cancer, were awarded $4.7 billion by a St. Louis jury.
You see, Johnson & Johnson has been able to keep their internal documents private for years, labeling them “confidential.” This new report sheds light on information that has not been made available to the public before – including internal documents, research results, and trial testimony – which tells a chilling tale. According to Reuters:
“[The information] shows that from at least 1971 to the early 2000s, the company’s raw talc and finished powders sometimes tested positive for small amounts of asbestos, and that company executives, mine managers, scientists, doctors and lawyers fretted over the problem and how to address it while failing to disclose it to regulators or the public.”
It goes on to say that the documents “also depict successful efforts to influence U.S. regulators’ plans to limit asbestos in cosmetic talc products and scientific research on the health effects of talc.” Reports from as far back as 1957 describe talc contaminated with fibrous tremolite, a recognized type of asbestos.
You can read these previously unreleased documents for yourself here.
Americans have been using Johnson and Johnson products since the 1880s, and Johnson’s Baby Powder (officially branded in 1893) has been used by mothers to help prevent chafing in diaper-aged children for well over a century. The Johnson & Johnson brand was established on the “Safety First” motto and is one of the only health and beauty companies in the world to put the company name on all of their products.
And for 125 years, that approach has worked. Johnson & Johnson is a massive company that has been trusted by consumers to be safe and effective. In addition to Johnson’s Baby Powder, the company also manufactures:
- Band-Aid
- Motrin
- Tylenol
- Benadryl
- Listerine
- Aveeno
- Acuvue
- Clean & Clear
- Neutrogena
- Rogaine
- Lubriderm
- and a host of other recognizable brands
Although Johnson’s Baby Powder accounts for only a small percent of their nearly $80 billion annual revenue, it is one of the most widely used personal care products worldwide. With one of the most recognizable fragrances in the world, the powder has been used for infants in diapers, as a feminine hygiene product, to prevent chafing, and as an all-purpose freshener.
And its users invariably inhale the carcinogenic, airborne powder.
Johnson & Johnson has since launched an aggressive campaign, attempting to stop one of the biggest financial slides in the company’s history. They’ve redesigned their website, revising a section called “Talcum Powder and Cancer” which used to say that “all talcum products have been asbestos free” to now say that guidelines state talc products should be free of asbestos. The website’s homepage now features a message from CEO Alex Gorsky about talc safety. Gorsky even appeared on CNBC’s Mad Money last night in an interview with the show’s host, Jim Cramer.
“What’s really important to focus on is not to select just one document, one piece of evidence, but to look at the body of evidence in totality” Gorsky said on the show. “And when you do that, in this case, again, we remain very confident in the safety of our products, but more importantly, the actions of our people.”
But the evidence tells a different tale, in which J&J executives repeatedly made efforts to silence any studies that showed the powder to be harmful. In the 1970s, several studies and independent researchers found trace amounts of asbestos in Johnson & Johnson powders. But that didn’t stop a powerful spin machine from protecting the money-making giant.
In 1971, researchers from Mount Sinai Medical Center found asbestos in the lungs of people who had never worked with the mineral. They posited that talc powders, often contaminated with asbestos, may play a role and reported their findings to New York City environmental protection chief Jerome Kretchmer. A press conference was called, and an inquiry opened by the Food and Drug Administration (FDA).
The company issued the following statement:
“Johnson & Johnson takes great care to assure the purity of its products, even to the extent of mining and processing our own talc for use in baby powder. Our fifty years of research knowledge in this area indicates that there is no asbestos contained in the powder manufactured by Johnson & Johnson.”
Months after the statement was released, mineralogist Arthur Langer, of the Mount Sinai research team, wrote a letter to Johnson & Johnson. He informed them that he had found a “relatively small” amount of chrysotile asbestos in Johnson’s Baby Powder. Both the researchers and Kretchmer were added to the company’s list of “antagonistic personalities” a year later.
Meanwhile, Johnson & Johnson appeared to be cooperating with the FDA, sending samples of its talc to private labs for testing. The company sent the results of the tests to the FDA with a cover letter stating that the results “clearly show” no sign of chrysotile asbestos. An FDA document said that J&J provided “evidence that their talc contains less than 1%, if any, asbestos.”
But the information Johnson & Johnson shared with the FDA was incomplete. It excluded the results of testing on Shower to Shower by University of Minnesota professor Thomas Hutchinson. Professor Hutchinson found chrysotile in the popular powder that he described as “incontrovertible asbestos” in his lab notes.
The Reuters investigation found several more instances in which Johnson & Johnson deliberately omitted information or research results to present their products in a more favorable light. Still, the company had to acknowledge that there may be trace amounts of asbestos in their powders.
They launched studies and lobbied the FDA, citing a “large safety factor” for talc containing less than one percent asbestos. They claimed that the exposure was still inside OSHA’s workplace exposure limits and shouldn’t be an issue for consumers. An FDA official named Dr. Shaffner was quoted as saying the idea was foolish, because “no mother [is] going to powder her baby with one percent of a known carcinogen irregardless [sic] of the large safety factor.”
And why would they? Why would any of us continue to use a product that’s been shown to cause cancer? The answer is: we wouldn’t. And that’s why Johnson & Johnson has gone to such great lengths to keep this information from both federal regulators and consumers. What’s worse, Johnson’s Baby Powder only accounts for half a percent of the company’s annual revenue. The lies and deceit are protecting an extremely small part of the pharmaceutical giant’s empire.
In late 2022, CEO Alex Gorsky stood in front of a camera on national television and told the American public that he believes “unequivocally” that J&J powders don’t contain asbestos. He went on to say that we needed to look at the body of evidence “in totality,” claiming that any evidence that showed the presence of asbestos could be blamed on varied testing methods or statistical outliers. But is that true?
Lawyers defending Johnson & Johnson in court have given a plethora of wild excuses for the presence of asbestos in their talc samples. “This sample was intended for industrial use” they claim. “That type of asbestos is harmless.” They’ve even suggested that some talc samples had been contaminated by “background” asbestos. That the talc was pure and must have been contaminated somehow during testing.
But if that’s true, why has the company kept the information from the public for so long? Why, as Reuters discovered, have they made such a concerted effort to influence policy and regulation involving talc purity and testing? In her ruling against J&J, Middlesex County Superior Court Judge Ana Viscomi said:
Providing the FDA favorable results showing no asbestos and withholding or failing to provide unfavorable results, which show asbestos, is a form of a misrepresentation by omission.”
To be clear, “misrepresentation by omission” means lying. But this wasn’t the first time that a major pharmaceutical company lied about product safety, and it won’t be the last.
To learn more about what’s REALLY going on behind the scenes, check out Propaganda Exposed!
A confidential memo to Johnson & Johnson managers in the baby products division shows the precise strategy used to deal with the issue of talc powder and cancer:
“Our current posture with respect to the sponsorship of talc safety studies has been to initiate studies only as dictated by confrontation. This philosophy, so far, has allowed us to neutralize or hold in check data already generated by investigators who question the safety of talc. The principal advantage for this operating philosophy lies in the fact that we minimize the risk of possible self-generation of scientific data which may be politically or scientifically embarrassing.”
Belviq
At the start of 2020, the FDA issued a warning that the weight loss drug lorcaserin hydrochloride (sold under the brand name Belviq) was associated with an increased risk of cancer. The drug was designed to increase feelings of fullness. While the studies are ongoing, the initial data suggests a connection between the diet pill and cancer.
Researchers don’t yet know the mechanism and can’t say definitively whether there is correlation or causation, but one thing is clear: people taking the weight loss drug are more likely to develop cancer. The research is just recently underway, even though the drug was approved by the FDA way back in 2012.
The decision was a relatively controversial one, given that lorcaserin only showed a 3.3% improvement over the placebo group. There was also concern that lorcaserin could cause heart valve issues, but there was not enough data done to prove it one way or the other. Still, the FDA’s Endocrinologic and Metabolic Drugs Advisory Committee voted 18-4 – with one abstention – in favor of approving the drug.
It was also not the first time that lorcaserin has been before an FDA review panel.
In 2010, Arena pharmaceuticals, the creators of Belviq, went before the FDA committee but was denied approval by a vote of 9-5. The committee members were asked to determine if the medical benefits of the drug outweighed the potential risks.
These members believed that the benefit of the drug was minimal, with less than half of all patients achieving at least a 5% loss in body weight. They were also concerned about data from animal studies that suggested an increased risk of tumors in rodents who received the drug.
The committee recommended in 2010 that the drug not be approved, citing small benefits and potential risks for cancer and valvulopathy (disease of the heart valves). They recommended further research to prove the safety and efficacy of the drug.
Just two years later, Arena brought the same drug in front of the committee. There was still concern about heart valve disease (and several panelists recommended that patients taking Belviq receive regular echocardiograms as a precaution). And the link between the drug and tumors in rats was waved off as something that should be of no concern in humans.
Chantix
In 2021, Pharmaceutical giant Pfizer expanded its recall of anti-smoking drug Chantix after testing found high levels of carcinogenic material in the pill. The leading prescription for smoking cessation, Chantix is prescribed to over half a million people each year and accounts for $1.1 BILLION in revenue for Pfizer each year.
While commercial cigarettes are undoubtedly unhealthy, the market for pharmaceutical cessation products is enormous. So is the money spent on advertising. Pfizer spends over $200 million a year to promote the drug (which is over 400% more expensive than it was in 2008).
As it turns out, patients looking to improve their quality of life and reduce their cancer risk by quitting cigarettes may have been ingesting a potent carcinogen instead. The cancer-causing agent in question – nitrosamines – are part of a large group of genotoxic chemical carcinogens that can be found naturally in some dip and chewing tobacco products.
Unfortunately, the issue of Pfizer making millions of dollars by selling unsafe drugs isn’t new. Pfizer has paid out BILLIONS in fines and settlements for promoting unsafe products, pushing off-label use on doctors, and lying about the safety or efficacy of their products.
In 2009, Pfizer (and its subsidiary Pharmacia & Upjohn Company) paid $2.3 BILLION to settle criminal and civil liabilities for illegal promotion of their pharmaceutical products. The amount included payment of more than $102 million in civil settlement to six whistleblowers of the company’s fraudulent practices.
Bextra, an anti-inflammatory drug that was withdrawn from the market in 2005 due to safety concerns, was marketed by the company for various off-label uses. The company also illegally promoted several other drugs, including antipsychotic drug Geodon, antibiotic Zyvox, and anti-epileptic drug Lyrica. Healthcare providers received payments for prescribing these drugs to patients for off-label use… literally bribing doctors to prescribe untested and unapproved drugs to patients.
False claims were submitted to government healthcare programs, allowing them to bypass the insurance programs. Pfizer had to pay approximately $1 BILLION to Medicare, Medicaid, and other government insurance programs under the settlement.
And those are just a few examples.
In 1993, anti-seizure drug gabapentin was widely prescribed for off-label uses such as treatment of pain and psychiatric conditions. Courts ruled that Pfizer utilized propaganda campaigns, paid for favorable research and coverage, and suppressed unfavorable research regarding the drug. Several regulatory bodies found the drug ineffective for the associated ailments, and Pfizer paid $430 million in one of the largest settlements to resolve criminal and civil health care liability charges.
A “whistleblower suit” was filed in 2005 against Wyeth, which was acquired by Pfizer in 2009, alleging that the company illegally marketed sirolimus (Rapamune) for off-label uses, targeted specific doctors and medical facilities to increase sales of Rapamune, tried to get transplant patients to change from their transplant drugs to Rapamune, and specifically targeted African-Americans.
According to the whistleblowers, Wyeth also provided doctors and hospitals that prescribed the drug with kickbacks such as grants, donations, and other money. In 2013, the company pleaded guilty to criminal mis-branding violations under the Federal Food, Drug, and Cosmetic Act. By August 2014, it had paid $491 million in civil and criminal penalties related to Rapamune.
In June 2010, health insurance network Blue Cross Blue Shield filed a lawsuit against Pfizer for allegedly illegally marketing drugs Bextra, Geodon, and Lyrica. Blue Cross alleged that Pfizer used kickbacks and wrongly persuaded doctors to prescribe the drugs. According to the lawsuit, Pfizer handed out ‘misleading’ materials on off-label uses, sent over 5,000 doctors on trips to the Caribbean or around the United States, and paid them $2,000 honoraria in return for listening to lectures about Bextra.
An internal marketing plan revealed that Pfizer intended to train physicians “to serve as public relations spokespeople.” The case was settled in 2014 for $325 million. Fearing that Pfizer was “too big to fail” (and that prosecuting the company would result in disruptions to Medicare and Medicaid), federal prosecutors instead charged a subsidiary of a subsidiary of a subsidiary of Pfizer, shielding them from virtually all financial responsibility.
In 2013, Pfizer agreed to a $964 million settlement for selling insulation laden with asbestos.
That same year, Pfizer withdrew “between $400,000 and a million dollars” worth of ads from Harper’s Magazine following an unflattering article on their depression medication.
In 1994, Pfizer agreed to pay $10.75 million to settle claims by the United States Department of Justice that the company lied to get approval for defective heart valves that killed roughly 500 people.
In 1996, an outbreak of measles, cholera, and bacterial meningitis occurred in Nigeria. Pfizer representatives and personnel set up a clinical trial and administered the experimental antibiotic trovafloxacin to approximately 200 children. Local officials reported that more than fifty children (over 25%) died in the experiment, while many others developed mental and physical deformities.
We could continue, but you probably see the pattern here. Pfizer doesn’t care if their products are safe. They’re willing to bribe doctors and silence the opposition to sell more products. They’re willing to KILL CHILDREN to make a buck.
Editor’s Note: This article was initially published in 2023 and has been updated in 2025.



















We need to take responsibility for our physical wellbeing and not take these poisonous drugs and vaccines sold by these criminal organizations. It’s easier for us to control our own behavior than try to make a criminal act morally responsible.
Great point, well stated.
Many years ago I watched your series “The Truth About Cancer”. It started my “awakening” process. It changed how I viewed doctors and what they prescribe. I have had many friends get angry with me when I have told them what I learned from you. I also have had a few friends listen. I was so impressed with that free series that I actually bought it. That series changed what I do for my own health. I think it’s time for me to rewatch it and get a refresher. Thank you Ty and Charlene for all you do!
Any I formation on Keytruda?
PLEASE add closed captioning to your series. Lots of older people, including myself and my husband, CANNOT understand the words in these videos. Thanks, Polly
Why doesn’t someone expose the chemicals in soda and make it a big deal. It causes cancer and other problems.
I would like to know about Prolia. I took this drug for 3 years. My PCP and Endocrinologist said this was a very great drug with a lot of good outcomes for people using it for Osteoarthritis. It almost killed me. If anyone has had a negative reaction or severe side effects, I would like too know.
Good for you guys, Ty and Charlene. Your work is excellent, exposing the horrible
abuses to innocent human beings by drug companies and the FDA. Who is like God,
no one is like God. The Globalists don’t think so. Just you wait Henry Higgins, just you
wait and see.
doctors are so highly educated beyond intelligence if they were intelligent they would stop doing what they are doing practicing in medicine they can practice on themselves not me