Editor’s Note: This article first appeared in the April 2016 edition of TTAC’s member newsletter.
The year 2015 was a busy one for me, as I spent many months traveling the world in search of everything there is to know about cancer – and more importantly, the many ways in which this prominent disease is being successfully treated throughout the world.
During my travels, I came across a revolutionary approach to cancer treatment that, in my humble opinion, just might be one of the most promising cancer therapeutic yet to come along in the 21st century.
It’s known as Rigvir® virotherapy, and an astute team of scientists, researchers, and doctors in the European country of Latvia are successfully using it right now to treat many different types of cancer. Among the many reasons why I believe Rigvir® is so promising is the fact that it’s shown to be extremely safe and 100% natural, derived from beneficial viruses that live naturally inside the human body. Rigvir® is also highly effective at treating many forms of cancer, which I’ll expound upon later.
Similar in function to the probiotic bacteria that populate the small intestine, the viruses from which Rigvir® is derived are recognized as beneficial pathogens that support the body’s innate immune system. These valuable viruses are both oncotropic and oncolytic, meaning they selectively target and destroy cancer cells. They do this without harming healthy cells, which can’t be said about conventional therapeutics such as chemotherapy and radiation.
I found the premise behind Rigvir® virotherapy as an efficacious cancer treatment so fascinating that I decided to conduct a series of interviews with the incredible minds behind its development. I also took the time to listen to a number of powerful life stories from people whose lives have been forever changed by this amazing treatment, which are nothing short of inspirational.
Discover the Amazing Healing Power of Rigvir®
The idea that viruses can act as helpful scavengers to root out infection and disease isn’t necessarily new. Saint Peregrine, the Roman Catholic patron saint of cancer patients who lived from 1265-1345, is said to have overcome an ulcerative growth on his leg as a result of a mystery virus that lived inside his body. Fast-forward about 600 years and the word “virotherapy” made its first appearance in the PubMed database around 1960.
It was around this time that emerging research began to toy with the idea that viruses could potentially be used as virotherapeutics to alter the way cancer grows and spreads. This hypothesis turned out to be correct – viruses directly affect the growth and spread of cancer cells. And now, thanks to the blood, sweat, and tears of scientific progressives in Latvia, we finally have access to the world’s first science-based virotherapeutic in the form of Rigvir.®
Rigvir® has its origins in the Latvian capital of Riga, where scientists there began looking at it as a possible virotherapeutic as far back as the late 1960s. A shorthand derivative of “Riga Virus,” Rigvir® is based on a virus known as Echo-7 that was first extracted from the gut microflora of healthy children as part of early medical testing for the polio vaccine.
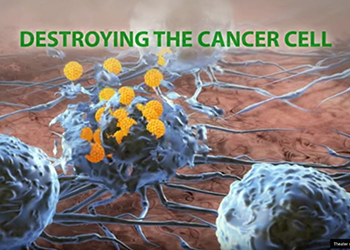
The Rigvir® virus enters a cancer cell and reproduces until the cell is destroyed
After this testing was completed, scientists began to take a closer look at the many isolated viral samples they had on hand, discovering in the process that Rigvir® possesses unique anti-cancer potential. They learned that Rigvir® is able to gain unique entry into cancer cells where it perpetually reproduces until those cells are eventually destroyed.
When Rigvir® is introduced into a cancer patient’s body via intramuscular injection, it very rapidly activates the body’s natural immune response, as well as the immune cells found inside the lymph nodes. It then actively and aggressively seeks out cancer cells, attaching to their surfaces for penetration, replication, and ultimate destruction.
Clinical testing has shown that Rigvir® acts quickly and powerfully in eradicating cancer cells throughout a patient’s body, showing near-immediate effects. Although it’s only been approved in two countries (Latvia and Georgia), for use in the treatment of melanoma, Rigvir® has also shown incredible success in the treatment of:
- Colorectal cancer
- Kidney cancer
- Bladder cancer
- Prostate cancer
- Stomach cancer
- Pancreatic cancer
- Uterine cancer
- Lung cancer
- Several types of sarcoma cancer
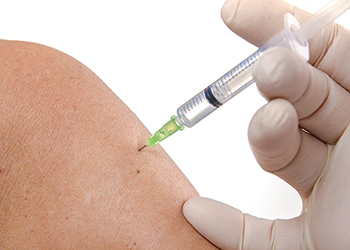
The Rigvir® virus is injected into the muscle. From there it activates the body’s immune response, including immune cells in the lymph nodes
What sets Rigvir® apart from many of the other cancer therapies out there is its purity. The only thing in a vial of Rigvir® is the virus and purified saltwater – that’s it! None of the preservatives, heavy metals, or adjuvants commonly found in vaccines and pharmaceuticals are present, which makes Rigvir® exceptionally safe and virtually side effect-free.
“[Rigvir®] contains the virus and almost nothing else except for normal saline for injection,” says Professor Ivars Kalvins, PhD, a scientist, inventor, and Director of the Latvian Institute of Organic Synthesis, which produces Rigvir® treatments for patients.
“This means that by administration of this (treatment), the body is not exposed to anything else, no additives, no toxic substances, only the live virus that upon entry into the body fluids starts searching for tumour cells – tumour cells that the virus eventually infects and kills.”

Dr. Ivar Kalvins (left) helped pioneer the development of Rigvir®
Dr. Ivars Kalvins, who helped pioneer the development of Rigvir,® was one of three finalists for the European Medicine Award. Dr. Kalvins is proud of Rigvir’s® successful track record as seen over the past 12 years since it first gained commercial approval.
For my docu-series, The Truth About Cancer: A Global Quest, I got a chance to speak with Dr. Kalvins about the nature of Rigvir® and how it works. He told me that, in addition to its immunomodulating efficacy, Rigvir® is characterized as being selectively toxic, meaning it doesn’t harm healthy cells.

Rigvir® has demonstrated numerous benefits without the harmful side effects of many coventional cancer treatments
Most other drugs, he told me, “are also killing healthy cells, not only the cancer cells. The only way to overcome this is to use living species like viruses. If you can find the virus [that specializes at] finding cancer cells and penetrating into these cells using the machinery, the factory, of these cells to reproduce themselves, this is exhausting the resources of (cancer) cells, and cells are dying out. This is the new approach. There is only one medicine in the world on the market that uses live virus – Rigvir.®”
Rigvir® truly is a one-of-a-kind cancer treatment, and the first ever virotherapeutic in the world to gain commercial approval. And I’m excited to say that it’s helping people from every corner of the globe overcome cancer, including late-stage cancers that the medical industry says are “incurable.”
Real-Life Testimonies from Patients Cured of Cancer with Rigvir®
One sweet Ukrainian lady I interviewed on my trip to Latvia is Khrystyna Yakonvenko. She told me her amazing story of overcoming stage IV melanoma with Rivgir.® Stage IV melanoma, as you may already know, is considered by the conventional medical system to be incurable due to the fact that cancer cells have already spread from the lymph nodes to vital organs and soft tissue.
“It all started in the end of 2012,” Khrystyna explained. “When I contacted my doctor, he diagnosed melanoma of the fourth stage, with metastasis of the liver. They prescribed the palliative chemotherapy, and … I completely trusted our Ukrainian doctors and the methods they were using, and I trusted this palliative chemotherapy – I simply didn’t realize the effect of this diagnosis completely and entirely in that moment.”
When I asked Khrystyna how long she was expected to live, “maximum of about six months,” was her response, according to her doctors’ assessment. But Khrystyna didn’t let this diagnosis get the best of her.

Khrystyna Yakonvenko wipes away a tear as she tells how she almost died from Stage 4 melanoma prior to receiving Rigvir® therapy
“I was not feeling afraid; I was not falling into panic,” she told me, as tears began to form. “Simply, I understood that I had no right to live it, and I had to fight. When I first came to the Rigvir® therapy center, the doctors didn’t say that, yes, we will do it – they said, we will try, because the stage is late. And I think that sometimes on the earlier stages, on the initial stages, people who have this very scary diagnosis, they sometimes by themselves, they lose hope, they stop fighting, and they simply leave it.
But sometimes there are people who, even at the late stages, they continue to fight, they continue to find the way out of the situation, and in this case, the disease is simply just over,” Khrystyna explained, noting that her stage IV melanoma is now completely gone.
Zoya Sokolova from Russia had a similar story to tell, except hers started off much differently. After being diagnosed with Stage 3 sarcoma, Zoya decided to go the conventional treatment route, only to end up on the verge of death! Fortunately for Zoya, her family intervened and took her to get Rigvir® Virotherapy before it was too late.
The diagnosis was very sudden for me. It was after a very strong stress, and then after a month and a half I was diagnosed with a third-stage cancer. After the surgery, I had my chemotherapy, then I signed (up for) another six of chemotherapy, and a full course of radiotherapy.
My condition allowed only for chemotherapy to be handled, and after the fourth, I wasn’t able to stand up from the bed. I became a bed patient. Before these courses of chemotherapy, I was told about the (Rigvir®) center, about this treatment, but I believed in our doctors and their treatment methods, so I decided to follow that path.
It seemed that they wanted for me to be treated, but they couldn’t give a warranty. They couldn’t say for sure that [they] would treat this disease. And that moment when I couldn’t stand up from the bed on my own completely, I was so weak, and my relatives they decided to take a van to make a bed for me, and simply drive me to Riga to the center.
Before coming to Riga, I got a blood test and a complete observation for the doctors here to have the full picture of my condition. When the doctor saw my blood test, she was really astonished because the other blood test was lower than for a live person. She was astonished how I managed to get here staying alive.
The doctor said exactly, ‘I’m not asking how you got here, I’m asking how are you still alive with this blood test?’ From my feelings at that moment, I realized that I would not survive, and I felt how my body is failing from day to day. The doctors recommended that I take certain measures, to take certain indications… before even taking Rigvir,® to boost my immune system, to reinvigorate and repair my immune system and my body so I could be ready for Rigvir.® And after a couple of weeks, I started to take Rigvir.®
It was really my family who made the decision because, as for me, I was so weak, I was so done, that I simply took this and I was ready to say goodbye to the life, [but] my family made the decision. I simply accepted their decision and I thought, what shall be shall be, [so] I followed them.
After following the indication of the doctors for boosting my body and my immune system [for] two weeks, I was able to stand up from the bed and walk around the house and move, so I was feeling better. After a month of already taking Rigvir® virotherapy, my condition changed entirely. I was now even able to walk around, stand up from the bed, I was even able to drive myself to Riga to receive the therapy.

Zoya Sokolova started chemotherapy treatments with a group of women who all rejected Rigvir® treatment as an option. Zoya is the only cancer survivor from the group
Zoya’s story is a tearjerker, but she was the fortunate one. The other women with whom she had been receiving treatment for similar cancers while still in Russia weren’t so fortunate, as all of them rejected the Rigvir® treatment in favor of chemotherapy and radiation. And all of them paid for this decision with their lives.
“When I had my surgeries, I had them with the same women in the same room, so we were following the same path, and met on the same days for chemotherapy. And when I started Rigvir,® even though I had only four (regimens of) chemotherapy, not the full course, they continued this course, and as I was getting better with Rigvir,® they on chemotherapy were starting to feel worse and worse,” recounted Zoya.
When I started to receive Rigvir® therapy, very quickly I became a very healthy person. I started to travel, I started to have a lot of energy and I called them and I recommended that they start this treatment. But for some reasons they refused, and now they’re all gone.
It’s a pity that a lot of great people are gone now. But I’m happy, I’m healthy, and I can’t say that at the beginning I didn’t trust this method, simply I didn’t know and I was so weak. But now, currently, I don’t have anymore disabilities, I’m a completely healthy person and I’m so thankful to the people who have helped me here.
I can’t even begin to tell you how much joy was brought to my heart knowing that these two lovely women are still with us today and living vibrant lives thanks to Rigvir® Virotherapy. The same can be said for many other patients like them who I personally met in Latvia, including:
- Karlis Venskus, who was cured of Stage 4 stomach cancer with Rigvir®
- Egidijus Kazlauskis, who was cured of Stage 3 melanoma with Rigvir®
- Gunars Strazdinsh, who was cured of Stage 3 small cell lung cancer with Rigvir®
- Ruslan Isayev, who was cured of Stage 3 skin melanoma with Rigvir®
- Svetlana Sheferova, who was cured of Stage 3 melanoma with Rigvir®
FDA Approves GMO Knockoff of Rigvir® Cancer Treatment
You’re probably asking yourself right about now – how can I access Rigvir® here in the United States for myself or a loved one? The unfortunate answer is that Rigvir® isn’t approved for use in the U.S., and is only available in Latvia, Mexico, the Bahamas, Germany, and the country of Georgia.
The reason for this is a systemic bias within the U.S. Food and Drug Administration (FDA) that prevents life-saving cancer treatments that actually work from ever being commercially approved. I cover this conspiracy in considerably more depth in my docu-series, but suffice it to say that the cancer industry has a stranglehold on the ways in which cancer can be legally treated here in the States.
To make matters worse, the FDA did recently approve another form of oncolytic virotherapy known as “Imlygic”™ (talimogene laherparepvec) that, like Rigvir,® is the first of its kind to be approved for use in the treatment of melanoma. But unlike Rigvir,® Imlygic™ is genetically-modified (GMO) and non-selectively toxic, meaning it harms both benign and malignant cells.
Even worse is the fact that Imlygic™ isn’t even all that effective, nor does it root out advanced-stage melanomas that have already spread to vital organs and other soft tissue. Imlygic,™ which is made of GMO herpes virus, can also infect patients with herpes, as admitted by the FDA in a recent press announcement.
A mere 16.3 percent of melanoma patients treated with Imlygic™ in a multi-center clinical trial saw any improvements at all from the drug’s use, according to the FDA. And the drug did not show benefits among patients whose melanomas had already spread to the brain, bone, liver, lungs, or other internal organs.
According to the Imlygic website, “IMLYGIC™ has not been shown to improve overall survival or have an effect on visceral metastases” (meaning cancer of internal organs)
Imlygic™ is also loaded with various chemical additives like disodium hydrogen phosphate dihydrate (pH balancer) and sorbitol (sugar alcohol) that have a questionable safety profile when injected directly into muscle tissue. Rigvir,® as I mentioned earlier, is made up of just the natural virus and saltwater.
Imlygic™ pales in comparison to Rigvir® on every level from its safety to its effectiveness, not to mention the fact that Imlygic™ is made from a recombinant (genetically engineered) herpes virus that exists nowhere in nature, while Rigvir® is made from a naturally-occurring virus that is simply an extension of the body’s own native immune system.
We can’t keep shunning effective treatments like Rigvir® in favor of questionable ones like Imlygic,™ simply because drug companies (in this case Amgen), have enormous amounts of cash to shell out to the FDA in exchange for a regulatory stamp of approval. The system is broken, and tens of thousands, and even millions, of people are needlessly dying from preventable diseases as a result.
It is my sincere hope that someday our country will recognize other forms of medicine besides just pharmaceuticals and surgery, and that cancer patients who wish to undergo treatments that fall outside the norm will be able to do so, within our own country, legally, and with full support of health insurance providers.
Article Summary
Doctors are successfully using virotherapy to treat and cure many different types of cancer.
Virotherapy first appeared in the PubMed database around 1960.
Rigvir® is able to gain unique entry into cancer cells where it perpetually reproduces until those cells are eventually destroyed.
Rigvir® is the first virotherapeutic to gain commercial approval.
Unlike American knockoffs, Rigvir® is selectively toxic, meaning it doesn’t harm healthy cells.



















Hi,
I want to know which medicine will work for Triple negative stage 4.My mom is suffering from past three years.please please please help me.Is there cure for this TNBC?
Nithya,
The best advice we can give you is to consult with one of the doctors/experts that we interviewed in our Global Quest Series. Here is a link to get their information: https://thetruthaboutcancer.com/experts-info-sheet/
Wishing your mom all the best!
Please, talk about BCG therapy, for bladder cancer, is.that sure, effective??!!
Are you awhere of any program that would help us to get to a rigvir center? My husband has been told he has stage 4 lung cancer. He wants to fight it. Please!
My mom has has two diagnoses of lung cancer, though thankfully both times caught in Stage 0 or 1. She only needed surgery the first time and this time it was 5 rounds of radiation—we are still waiting to hear the results in the 3 month checkup. Really good nutrition is key to fighting this. There is an Asian mushroom supplement protocol that is shown to be quite effective for some lung cancers and you might want to also consider stem cell therapy. If you are in the US, Texas seems to have the most STC options due to recent legislative changes there. Best of luck!
Yes where can we get rigvir. If in Mexico, where? I know people with cancer! Will there be anything orally to buy in the future. With this vaccine? Can we take a vaccine even if we are healthy as a preventative.
Hi Terry –
I believe the Rigvir therapy is offered at the Hope4Cancer clinic in Tijuana.
I’m also attaching the info about the Hope4Cancer clinic in Tijuana, MX if you’d like to reach out to them.
http://www.hope4cancer.com/
https://hope4cancer.com/schedule-a-call/
These are great questions, but it would be best if you reached out to a healthcare professional with your questions.
Unfortunately, we are unable to give any kind of medical advice. The best advice we can give you is to consult with one of the doctors we interviewed in the Global series.
Please note that we are not able to select an expert for you.
We’ve created a page with the experts’ contact info as it was available to us.
Here’s the link to the actual webpage:
http://thetruthaboutcancer.com/experts-info-sheet/
We do have one more resource you may be interested in. During our Live Event 2017 series, Dr. Patrick Quillin suggested contacting The Institute of Functional Medicine to locate a practitioner in your area.
If you are interested, here is the link to search for a Functional Medicine Practitioner in your area: https://www.ifm.org/find-a-practitioner/
Hope this is helpful. Thanks for reaching out to us!
Sir:
Where is the source of this Rigvir in Mexico. Really URGENT
Ukegbu
Hi Okezie –
Thanks for reaching out to us. You can try consulting with the Hope4Cancer clinic in Tijuana – I believe they offer this therapy. If not they will be able to better guide you.
I’m attaching the info about the Hope4Cancer clinic in Tijuana, MX if you’d like to reach out to them.
http://www.hope4cancer.com/
https://hope4cancer.com/schedule-a-call/
Hope this is helpful. Wishing you endless blessings and love!
Sir ,
Please suggest a treatment for stage 4 bone mets for my mom. She is going through immense pain.
Already taking ozone ,vitamin C and k2 .
Please suggest .
Thanks
Hi Ramya –
We appreciate you reaching out to us about this.
I’m so sorry to hear about your mother – I’ll be sure to pray for her.
Unfortunately, we are unable to give any kind of medical advice. The best advice we can give you is to consult with one of the doctors we interviewed in the Global series.
Please note that we are not able to select an expert for you.
We’ve created a page with the experts’ contact info as it was available to us.
Here’s the link to the actual webpage:
http://thetruthaboutcancer.com/experts-info-sheet/
We do have one more resource you may be interested in. During our Live Event 2017 series, Dr. Patrick Quillin suggested contacting The Institute of Functional Medicine to locate a practitioner in your area.
If you are interested, here is the link to search for a Functional Medicine Practitioner in your area: https://www.ifm.org/find-a-practitioner/
I’m also attaching the info about the Hope4Cancer clinic in Tijuana, MX if you’d like to reach out to them.
http://www.hope4cancer.com/
https://hope4cancer.com/schedule-a-call/
Please also see the resources below in case they will be helpful to you.
From our beginning, we have supported charities that really help these cancer patients.
HealingStrong:
http://healingstrong.com/
https://www.facebook.com/healingstrong
HealingStrong supports MANY cancer patients and are amazing. HealingStrong, a nonprofit organization, is supported 100% by volunteers, who are experienced in using natural strategies. The organization’s mission is to educate, encourage, equip, and empower those seeking comprehensive natural strategies to heal strong and stay strong.
HealingStrong Connect Groups meet monthly in various areas of the U.S. and South Australia to link others interested in natural strategies, holistic protocols, and local resources. Their groups focus on mind, body and emotional healing based on Biblical promises, as they believe the God of the Bible is our healer. For more information on how to start a group, or become involved in one, please go to: http://www.healingstrong.org/groups, and like us on Facebook at http://www.facebook.com/healingstrong.
Cancer Crackdown is so awesome and helps patients without a lot of money get supplements and treatments and even helps them raise money:
http://cancercrackdown.org/
https://www.facebook.com/CancerCrackdown
Also, the Independent Cancer Research Foundation is another charity that we support, also known as Cancer Tutor. They have been a TREMENDOUS help for cancer patients and have a TON of life saving information they give out freely and they do real research into treatments for cancer that are outside of the toxic Big Pharma Big 3:
http://www.theicrf.com/
The best that we can suggest is to contact Healing Strong and Cancer Crackdown for any need in financial resources.
We also support Mary Beth’s “Nick Gonzalez Foundation” which is bringing Nick’s protocol to the people…which is one of the most powerful cancer killing protocols out there.
I hope this is helpful! Please don’t hesitate to reach out to us again if there’s anything else we can help with.
Wishing you endless blessings and love!
I have multiple myeloma for 13years keeping it in check for 12years naturally with Apricot kernel, green tea and a set natural diet however the last year I have eaten a lot of food with sugar.
My myeloma has reared its ugly head so I want to know what can I do or what is out there that can boost my management of myeloma
Hi Virginia –
We appreciate you reaching out to us about this.
I’m so sorry to hear about your health – I’ll be sure to pray for you.
Unfortunately, we are unable to give any kind of medical advice. The best advice we can give you is to consult with one of the doctors we interviewed in the Global series.
Please note that we are not able to select an expert for you.
We’ve created a page with the experts’ contact info as it was available to us.
Here’s the link to the actual webpage:
http://thetruthaboutcancer.com/experts-info-sheet/
We do have one more resource you may be interested in. During our Live Event 2017 series, Dr. Patrick Quillin suggested contacting The Institute of Functional Medicine to locate a practitioner in your area.
If you are interested, here is the link to search for a Functional Medicine Practitioner in your area: https://www.ifm.org/find-a-practitioner/
I’m also attaching the info about the Hope4Cancer clinic in Tijuana, MX if you’d like to reach out to them.
http://www.hope4cancer.com/
https://hope4cancer.com/schedule-a-call/
Please also see the resources below in case they will be helpful to you.
From our beginning, we have supported charities that really help these cancer patients.
HealingStrong:
http://healingstrong.com/
https://www.facebook.com/healingstrong
HealingStrong supports MANY cancer patients and are amazing. HealingStrong, a nonprofit organization, is supported 100% by volunteers, who are experienced in using natural strategies. The organization’s mission is to educate, encourage, equip, and empower those seeking comprehensive natural strategies to heal strong and stay strong.
HealingStrong Connect Groups meet monthly in various areas of the U.S. and South Australia to link others interested in natural strategies, holistic protocols, and local resources. Their groups focus on mind, body and emotional healing based on Biblical promises, as they believe the God of the Bible is our healer. For more information on how to start a group, or become involved in one, please go to: http://www.healingstrong.org/groups, and like us on Facebook at http://www.facebook.com/healingstrong.
Cancer Crackdown is so awesome and helps patients without a lot of money get supplements and treatments and even helps them raise money:
http://cancercrackdown.org/
https://www.facebook.com/CancerCrackdown
Also, the Independent Cancer Research Foundation is another charity that we support, also known as Cancer Tutor. They have been a TREMENDOUS help for cancer patients and have a TON of life saving information they give out freely and they do real research into treatments for cancer that are outside of the toxic Big Pharma Big 3:
http://www.theicrf.com/
The best that we can suggest is to contact Healing Strong and Cancer Crackdown for any need in financial resources.
We also support Mary Beth’s “Nick Gonzalez Foundation” which is bringing Nick’s protocol to the people…which is one of the most powerful cancer killing protocols out there.
I hope this is helpful! Please don’t hesitate to reach out to us again if there’s anything else we can help with.
Wishing you endless blessings and love!
So how does anyone in America get this drug if it isn’t available here? What do we do?
With all of this information (which is great and I’m so thankful for this site and all the time and effort you put into this) how do I know which way to go? My father in law is literally giving up. He has had so many different kinds of cancers for the last 10+ years and has managed to beat them but this bone cancer has taken a toll! Which doctor on your recommend list should I contact? Could you give me direction to go such as Mexico, Germany….?With so many article I don’t know what is best for him and what is best for my family and I to prevent it. I get it basically but is there a common or most recommended regimen for us? I’ve bought your books and watch your seminars! I read your emails daily but it’s an information overload. Thank you very much for everything
Sarah Ater
Hi Sarah –
We appreciate you reaching out to us about this.
I’m so sorry to hear about your father – I’ll be sure to pray for him.
Unfortunately, we are unable to give any kind of medical advice. The best advice we can give you is to consult with one of the doctors we interviewed in the Global series.
Please note that legally we are not able to select an expert for you.
We’ve created a page with the experts’ contact info as it was available to us.
Here’s the link to the actual webpage:
http://thetruthaboutcancer.com/experts-info-sheet/
We do have one more resource you may be interested in. During our Live Event 2017 series, Dr. Patrick Quillin suggested contacting The Institute of Functional Medicine to locate a practitioner in your area.
If you are interested, here is the link to search for a Functional Medicine Practitioner in your area: https://www.ifm.org/find-a-practitioner/
I’m also attaching the info about the Hope4Cancer clinic in Tijuana, MX if you’d like to reach out to them for the best direction.
http://www.hope4cancer.com/
https://hope4cancer.com/schedule-a-call/
Please also see the resources below in case they will be helpful to you.
From our beginning, we have supported charities that really help these cancer patients.
HealingStrong:
http://healingstrong.com/
https://www.facebook.com/healingstrong
HealingStrong supports MANY cancer patients and are amazing. HealingStrong, a nonprofit organization, is supported 100% by volunteers, who are experienced in using natural strategies. The organization’s mission is to educate, encourage, equip, and empower those seeking comprehensive natural strategies to heal strong and stay strong.
HealingStrong Connect Groups meet monthly in various areas of the U.S. and South Australia to link others interested in natural strategies, holistic protocols, and local resources. Their groups focus on mind, body and emotional healing based on Biblical promises, as they believe the God of the Bible is our healer. For more information on how to start a group, or become involved in one, please go to: http://www.healingstrong.org/groups, and like us on Facebook at http://www.facebook.com/healingstrong.
Cancer Crackdown is so awesome and helps patients without a lot of money get supplements and treatments and even helps them raise money:
http://cancercrackdown.org/
https://www.facebook.com/CancerCrackdown
Also, the Independent Cancer Research Foundation is another charity that we support, also known as Cancer Tutor. They have been a TREMENDOUS help for cancer patients and have a TON of life saving information they give out freely and they do real research into treatments for cancer that are outside of the toxic Big Pharma Big 3:
http://www.theicrf.com/
The best that we can suggest is to contact Healing Strong and Cancer Crackdown for any need in financial resources.
We also support Mary Beth’s “Nick Gonzalez Foundation” which is bringing Nick’s protocol to the people…which is one of the most powerful cancer killing protocols out there.
I hope this is helpful! Please don’t hesitate to reach out to us again if there’s anything else we can help with.
Wishing you endless blessings and love!
Too many of my relatives and friends have had their lives cut short by Cancer. A treatable and curable ailment that the NHS, English Law and Chemical Laboratory owners defiantly will not recognise, because their eye is on money.
Bring back the sanctity of life and care and love for humanity
My brother in law is dying of leukaemia & his bone marrow is not making platelets he is having blood transfusions weekly now & getting nose bleeding he is breaking out in cancers over his face a lump has appeared & grown to the size of an egg in one week?
Non of these treatments are available in Australia I can’t even get cannabinol oil for him
He’s going downhill fast
Hi Julie –
We appreciate you reaching out to us about this.
I’m so sorry to hear about your brother-in-law – I’ll be sure to pray for him.
Unfortunately, we are unable to give any kind of medical advice. The best advice we can give you is to consult with one of the doctors we interviewed in the Global series.
Please note that we are not able to select an expert for you.
We’ve created a page with the experts’ contact info as it was available to us.
Here’s the link to the actual webpage:
http://thetruthaboutcancer.com/experts-info-sheet/
Also, during our Live Event 2017 series, Dr. Patrick Quillin suggested contacting The Institute of Functional Medicine to locate a practitioner in your area.
If you are interested, here is the link to search for a Functional Medicine Practitioner in your area: https://www.ifm.org/find-a-practitioner/
I’m also attaching the info about the Hope4Cancer clinic in Tijuana, MX if you’d like to reach out to them.
http://www.hope4cancer.com/
https://hope4cancer.com/schedule-a-call/
Please also see the resources below in case they will be helpful to you.
From our beginning, we have supported charities that really help these cancer patients.
Cancer Crackdown is so awesome and helps patients without a lot of money get supplements and treatments and even helps them raise money:
http://cancercrackdown.org/
https://www.facebook.com/CancerCrackdown
Also, the Independent Cancer Research Foundation is another charity that we support, also known as Cancer Tutor. They have been a TREMENDOUS help for cancer patients and have a TON of life-saving information they give out freely and they do real research into treatments for cancer that are outside of the toxic Big Pharma Big 3:
https://thetruthaboutcancer.com/independent-cancer-research-foundation/
The best that we can suggest is to contact Cancer Crackdown for any need in financial resources.
We also support Mary Beth’s “Nick Gonzalez Foundation” which is bringing Nick’s protocol to the people…which is one of the most powerful cancer-killing protocols out there.
I hope this is helpful! Please don’t hesitate to reach out to us again if there’s anything else we can help with.
Wishing you endless blessings and love!
Where in Germany can you get Rigvir treatment?
Hi, have you got a contact address for the Rigvir Clinic in Latvia?
Is Rigvir treatment used for prostate cancer bone metastases?
What is the best treatment to try for prostate cancer bone metastases?