Ketogenic diets – also known as keto diets, or keto – are the topic of heavy conversation these days. A typical keto diet recommends getting 75% of calories from healthy fats, 20% from protein, and only 5% from carbs.
On Google, searches for keto now outrank those for paleo.1 Keto recipe books rank high on bestseller lists and believe it or not, high-fat snacks have become a flourishing business.
It may surprise you to learn that what we know today as the keto diet was developed as a clinical tool just under a hundred years ago. It all began in the early 20th Century, when patients with various illnesses were seen to benefit from short fasts.2
For instance, doctors noticed that children with epilepsy stopped having seizures after two days of absolute fasting at a point when their bodies would have been forced to enter a physiological state known as ketosis. This is believed to be an evolutionary adaptation to help us through times when food is scarce.
Promisingly, fasting was also found to have positive effects on their body fat, blood sugar, cholesterol, and hunger levels.
In 1921, Dr. R.M. Wilder at the Mayo Clinic proposed a diet for the treatment of epilepsy – which he referred to as a ketogenic diet – in which most of the calories were derived from fat, mimicking the biochemical changes of fasting.2 Today, children resistant to anti-epileptic drugs are still advised to follow keto diets to prevent seizures, always under the management of well-trained dietitians, of course. Indeed, according to the Epilepsy Society, the keto diet is considered to be a medical treatment.3
Scientists and clinicians have now begun to explore other potential health applications of the keto diet. Recent evidence suggests it can help to lower the risk of diabetes, heart disease, polycystic ovary syndrome (PCOS), neurological diseases, and cancer, among others.4
A recent news article describes the experience of Jan Vyjidak, who has been on the keto diet for nearly a decade, with the goal of better managing his health and improving his athletic performance.1
“It transformed my life,” he says. “It wiped out my constant hunger, and I was able to stop all medication for my asthma and psoriasis within six months.”
Strong evidence from the Prospective Urban Rural Epidemiology (PURE) study – a large, epidemiological study of over 1,350,000 individuals aged between 35-70 years from 18 countries – indicates that cutting down on refined carbs and increasing healthy fats can be beneficial to overall health.5
PURE study results showed that high carb consumption was associated with a greater risk of death, whereas consumption of total fat and individual types of fat were counterintuitively linked to a lower risk of death. Total fat and types of fat were not linked to a greater risk of developing cardiovascular disease, including heart disease.
Intriguingly, greater consumption of saturated fat was associated with a lower risk of stroke.
How Does the Keto Diet Work?
Normally our body uses sugar, also known as glucose, for fuel. However, we cannot make glucose in our body and only have about 24-36 hours’ worth stored in our muscle tissue and liver. Once glucose is no longer available from food sources and we’ve exhausted our internal stores, we begin to burn stored fat, or fat from our food.
Of course, if we consume more carbs than we need for our daily activities, the extra glucose gets stored as body fat and results in unwanted weight gain, with all its unwelcome health consequences.
A keto diet is based on the minimal consumption of carbs, especially refined carbs. Instead, adherents of this diet consume:
- Plenty of “healthy” high-fat foods such as grass-fed butter, avocado, olive, coconut, and palm oils, nuts, and seeds
- Moderate amounts of grass-fed meat, wild-caught fish, cage-free eggs, bone broth, organ meats, and cheese, and
- Carefully managed amounts of high-fiber, low-carb, non-irradiated, non-GMO vegetables such as cauliflower, broccoli, cabbage, spinach, zucchini, kale, and other leafy greens.
Foods rich in antioxidants are also allowed as long as they do not contain high levels of sugar. The specific details of a typical keto diet can be found here.6

Ketones are also capable of crossing the blood brain barrier and are an excellent fuel for our brain cells.
When our blood ketone levels reach a certain point, our body is said to enter a state of ketosis, typically three to four days after eliminating carbs or minimizing their consumption. Ketosis has been shown to help people lose weight and excess body fat rapidly – along with making many significant health gains – even while consuming lots of healthy fats and sufficient calories for their needs.
In order to maintain ketosis, carb intake must be restricted to less than 50 grams daily.
As protein can also be broken down into glucose in our body, moderate consumption of proteins is another aspect of this diet. Even fruit intake has to be carefully monitored, as many fruits have been bred for their sweetness and contain a lot of sugar.
Unfortunately, thanks to the widespread prevalence of the standard American diet (SAD) – characterized by high consumption of red meat, processed meat, pre-packaged foods, butter, fried foods, high-fat dairy products, refined grains, potatoes, corn, high-fructose corn syrup (HFCS), and high-sugar drinks – the incidence of obesity, insulin resistance, diabetes, cardiovascular disease, and many forms of cancer are skyrocketing in the U.S.
As we shall see, the keto diet might just be the magic bullet.
What is Cancer and What Causes It?
Cancer is not a single disease, but rather a group of diseases all of which share the common feature of abnormal cell growth. Cancer cells either stay put where they are formed or spread to other parts of the body. In the U.S. alone, it is estimated that nearly 2 million new cases of cancer will have been diagnosed in 2018, while over 600,000 people will have died as a consequence of this disease.7
Normal, healthy cells in our body grow and divide to form new cells, but only as and when our body needs them. When these cells grow old or become damaged, they die, and new cells typically take their place in an orderly fashion.
With cancer, however, this orderly process breaks down. Old and damaged cancer cells continue to live on when they should be dead, and new cells keep forming even though they are not needed. All of these cells, young and old, divide continuously without stopping to form masses of abnormal cells known as tumors.
Conventional medical thinking based on extensive scientific research over the past two to three decades states that many thousands of different types of errors in DNA – including so-called point mutations, amplifications, and deletions – are responsible for cancer formation.8
Some of these errors are believed to occur in genes in our DNA that control the way our cells function, especially how they grow and divide. These errors are believed to either turn on “oncogenes”, which promote cancer growth, or turn off “tumor suppressor genes”, which act to prevent cancer growth.
Metabolic Requirements for Cancer
Most of the DNA errors that lead to cancer appear to modify cellular metabolism in cancer cells, by targeting a dozen or so signaling pathways. Metabolism is a term used to describe chemical reactions that help to keep our cells and, by extension, our body, alive. Many of these metabolic changes are absolutely essential for cancer cell formation and survival.
For instance, rapidly dividing cancer cells have three fundamental metabolic requirements that they cannot live without.8
- They need to make a lot of ATP, and quickly, to support their high requirements for energy. Adenosine triphosphate, also known as ATP, is a compound that provides energy to drive hundreds of thousands of biochemical processes in living cells. Found in all forms of life, ATP is often referred to as the chemical energy “currency” that powers metabolic activity.
- Cancer cells need to keep making plenty of structural “macromolecules”, which are necessary for building more cancer cells as the tumor grows.
- Cancer cells need to carefully maintain their “redox status”. Redox status is the balance between oxidants and antioxidants. Oxidants, including free radicals and other “reactive” chemical species, are made continuously in every living cell as a byproduct of metabolic activities. Several antioxidant systems have evolved in our body to specifically counter the harmful actions of these oxidants.
For cells to maintain a healthy status, oxidants and antioxidants have to be in balance with each other. When this equilibrium is tilted toward an oxidized state, it leads to oxidative stress, in which an excess of oxidants can damage cellular structures and affect the health of the cell. Even cancer cells need to safeguard themselves against this.
Metabolism in Cancer: The “Warburg Effect”
Otto Heinrich Warburg
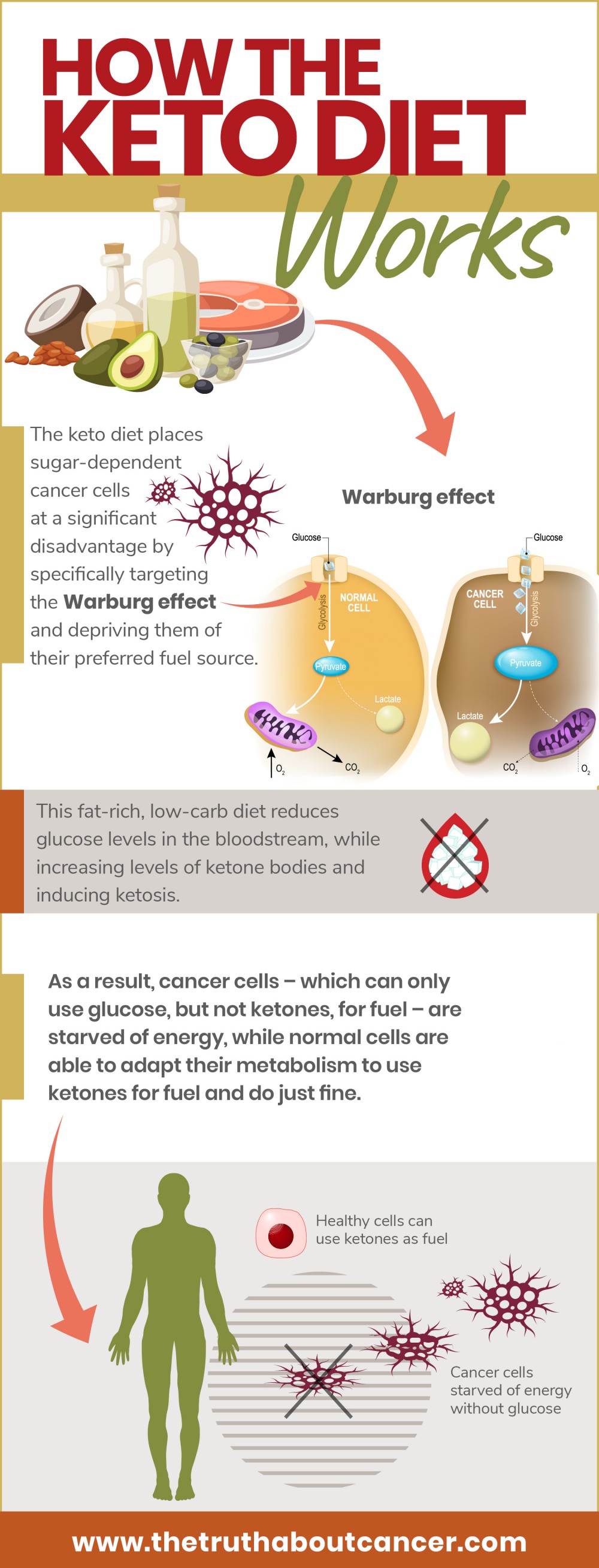
Back in the 1920s, Warburg conducted experiments in which he measured oxygen consumption and lactate production in tumor slices. Surprisingly, even in the presence of oxygen, rapidly growing tumor cells consumed glucose at a very high rate when compared to normal cells and produced lactate.9
Warburg proposed that this process, which he called “aerobic glycolysis”, was caused by the failure of mitochondrial metabolism in cancer cells. Further, he boldly claimed that aerobic glycolysis was in fact the actual cause of cancer cell formation.
In his 1956 paper in the scientific journal Science, Otto Warburg wrote:
What was formerly only qualitative has now become quantitative. What was formerly only probable has now become certain. The era in which the fermentation of the cancer cells or its importance could be disputed is over, and no one today can doubt that we understand the origin of cancer cells if we know how their large fermentation originates, or, to express it more fully, if we know how the damaged respiration and the excessive fermentation of the cancer cells originate.”
Every normal cell in our body produces several ATP molecules from just one glucose molecule – two via glycolysis, two via the tricarboxylic acid (TCA) cycle, and 34 via a process known as oxidative phosphorylation in mitochondria.
Mitochondria are the main power generators in our body’s cells and also play a prominent role in antioxidant activities and calcium buffering. The number of mitochondria present in any cell is directly related to its metabolic requirements and can range from a solitary mitochondrion to thousands of them. In fact, mitochondria enable cells to generate up to 15 times more ATP than they could otherwise!
Mitochondria generate reactive oxygen species (ROS) during their metabolic activities. In normal cells, the production of ROS and their elimination by antioxidants are kept in balance.10 Intriguingly, a higher incidence of errors, or mutations, in mitochondrial DNA have been observed in many human cancers, likely as a result of uncontrolled ROS production and oxidative stress.11
As a result, cancer cells are unable to make ATP via oxidative phosphorylation in mitochondria and instead resort to making ATP solely via glycolysis, even when oxygen levels are normal. This is the well-known “Warburg effect”.9
Although ATP production by glycolysis is quicker, it is much less efficient in terms of how much ATP can be generated per unit of glucose consumed. Also, glycolysis requires an abnormally high supply of glucose to meet the increased energy, macromolecular, and redox needs of cancer cells.
Some experts now feel that impaired energy metabolism may be the defining common factor in nearly all cancers, regardless of their origin.12,14 They believe that cancer, rather than being caused by errors in DNA, is fundamentally a metabolic disease caused by a disruption in a cancer cell’s ability to get the energy it needs from mitochondrial respiration like a normal cell would.
Further, these experts believe that DNA mutations, uncontrolled cellular growth, and other hallmarks of cancer are a consequence, not the cause, of impaired energy metabolism. They suggest that the poor rate of success in the “War on Cancer” has to do with mainstream medicine’s failure to recognize mitochondrial dysfunction as the underlying cause of cancer.
However, others still feel that DNA errors are indeed the main cause of cancer, and impairment of mitochondrial metabolism at best offers secondary survival advantages to cancer cells.
While this debate rages on, what is the relevance of impaired mitochondrial metabolism in cancer cells to the keto diet?
The simple answer is this – the keto diet places sugar-dependent cancer cells at a significant disadvantage by specifically targeting the Warburg effect and depriving them of their preferred fuel source.15 This fat-rich, low-carb diet reduces glucose levels in the bloodstream, while increasing levels of ketone bodies and inducing ketosis.
As a result, cancer cells – which can only use glucose, but not ketones, for fuel – are starved of energy, while normal cells are able to adapt their metabolism to use ketones for fuel and do just fine.
Further, both ROS production and oxidative stress are predicted to increase in cancer cells when confronted with the keto diet, damaging them as they try to make their ATP via their defective mitochondrial metabolism.
Based on this logic, the keto diet appears to be a safe, inexpensive, easily implementable, and effective approach to selectively target cancer cells. Promisingly, multiple studies show that the keto diet reduces tumor growth and improves survival in animal models of multiple cancers, along with enhancing the effects of other forms of anticancer therapy.11,15 Similarly, fasting has been shown to enhance responsiveness to chemotherapy, along with reducing some of the side effects in preclinical cancer therapy models.11
Let’s take a closer look at some of these studies.
A Closer Look at the Ketogenic Diet
It’s interesting that the “dietary religious fanatics” who believe everyone should eat a 100% plant-based diet seem to lose their mind when someone even suggests that the ketogenic diet might help reverse cancer, which many many studies indicate that it does.
Back in 2008, researchers tested the effects of a no-carbohydrate ketogenic diet (NCKD) relative to western and low-fat diets on prostate cancer growth in 75 mice with severe immunodeficiency.16
Despite consuming equal calories, NCKD mice were seen to lose up to 15% of their body weight relative to mice fed with either the low-fat or Western diet. Further, prostate tumor sizes were 33% smaller in NCKD mice relative to mice fed a western diet. NCKD mice also survived the longest of all.
Other studies have also shown the anticancer effects of a keto diet in combination with caloric restriction. For example, one such combination reduced the growth rate of a type of childhood cancer known as neuroblastoma and also prolonged survival in mice.17
Interestingly, these neuroblastoma cells showed low levels of mitochondrial metabolism as predicted by the Warburg effect.
Similarly, mice with a type of brain tumor known as malignant glioma lived longer when fed a keto diet, relative to control animals on a standard diet. The combination of the keto diet plus radiation therapy was seen to be additive, indicating that ketosis significantly enhances the anti-tumor effects of radiation.18
In a 2013 laboratory study, a keto diet alone was shown to significantly reduce blood glucose, slow tumor growth, and increase survival time by 56.7% in mice with systemic metastatic cancer.19 Metastatic cancers are cancers that can migrate from their place of origin to other areas of the body via the bloodstream or lymph.
Further, combining the keto diet with hyperbaric oxygen therapy was seen to lead to a further reduction in blood glucose, tumor growth rate, and a nearly 78% increase in mean survival time of these mice, relative to controls.
Hyperbaric oxygen therapy involves breathing pure oxygen in a pressurized room or tube and is a well-established treatment for decompression sickness after scuba diving. Hyperbaric oxygen therapy is also used to treat serious infections, bubbles of air in blood vessels, and wounds that don’t heal as a result of diabetes or radiation injury.
Similarly, in a 2015 study, mice receiving a combination of hyperbaric oxygen and dietary ketone supplementation showed a clear reduction in tumor growth rate and metastasis.20 Also, these mice lived twice as long as control animals. Based on these results, the study authors state that further investigation into the effectiveness of this combination therapy as a potential treatment for late-stage metastatic cancers is urgently required.
Dietary ketone supplementation has been shown to lower growth rate and survival of highly metastatic cancer cells implanted into mice and prolong their lifespan, even in the presence of high glucose.21
Last but not least, a meta-analysis of 12 studies on the effects of keto diets on mouse models of cancer concluded that “there was an overall tumor growth delaying effect of unrestricted KDs (keto diets) in mice”.22
Are There Adverse Effects of the Keto Diet?
Keto diets do show some side effects, including lethargy, nausea, and vomiting due to intolerance of the diet, especially in children, who may also exhibit low blood sugar and nausea.2 But more often than not, these are symptoms of “keto flu” and are generally a short-term reaction to the drastic change in diet.
In adults, GI discomfort is a common side effect due to the high fat content of the diet. A prospective pilot study on keto diets reported a significant, progressive increase in cholesterol levels after a year. In some instances, dramatic weight loss and liver dysfunction were seen.
Studies have also reported deficiencies in trace minerals like selenium, copper, and zinc in patients on keto diets. It’s important to remember that consuming enough mineral-rich foods and supplementation of trace minerals is likely necessary while on the diet.
Important Note: Remember, it’s advisable to talk to your doctor or a dietitian before beginning the keto diet, or any other diet, especially if you have been diagnosed with cancer. The fact is that different diet plans work well for different people, and your doctor or dietitian is best placed to help you decide whether the keto diet can help you reach your specific health goals.
Looking for more about the ketogenic diet?
- Discover why cyclical ketosis may be right for you.
- Learn how the ketogenic diet weakens cancer cells.
- Why all the hype about the ketogenic diet for cancer?
Article Summary
Ketosis is believed to be an evolutionary adaptation to help us through times when food is scarce.
Once we’ve exhausted our internal stores of glucose, we begin to burn stored fat or fat from our food.
The fat is broken down into ketones, which is excellent fuel for our brain cells.
Some experts believe that impaired energy metabolism may be the defining common factor in nearly all cancers.
The keto diet places sugar-dependent cancer cells at a significant disadvantage.
Promisingly, multiple studies show that the keto diet reduces tumor growth and improves survival in animal models of multiple cancers.
Remember to talk to your doctor or a dietitian before beginning the keto diet, or any other diet, especially if you have been diagnosed with cancer.



















It’s a very interesting concept, one to which I suscribe from the reading I’ve done.
One question Ihave is the liver produces glucose and maintains a ‘normal’ glucose level even when in ketosis. With this in mind, how does the blood glucose level drop thereby starving cancer cells?
CANCER ALSO HATES OXYGEN IN THE INTERNAL ENVIRONMENT
I have breast cancer i.e on my left breast, I believe what I have to do is change my diet & my lifestyle would help with the cancer that I have & I have faith in GOD. Thank you Mr.Ty Bollinger for what you’ve shared.
Hi Liti –
We’re so glad to hear of your positive outlook. What a blessing to have a hopeful attitude!
In case it’s helpful to you, we’ve created a page with the experts’ contact info as it was available to us.
Here’s the link to the actual webpage:
http://thetruthaboutcancer.com/experts-info-sheet/
We do have one more resource you may be interested in. During our Live Event 2017 series, Dr. Patrick Quillin suggested contacting The Institute of Functional Medicine to locate a practitioner in your area.
If you are interested, here is the link to search for a Functional Medicine Practitioner in your area: https://www.ifm.org/find-a-practitioner/
I’m also attaching the info about the Hope4Cancer clinic in Tijuana, MX if you’d like to reach out to them.
http://www.hope4cancer.com/
https://hope4cancer.com/schedule-a-call/
Please also see the resources below in case they will be helpful to you.
From our beginning, we have supported charities that really help these cancer patients.
HealingStrong:
http://healingstrong.com/
https://www.facebook.com/healingstrong
HealingStrong supports MANY cancer patients and are amazing. HealingStrong, a nonprofit organization, is supported 100% by volunteers, who are experienced in using natural strategies. The organization’s mission is to educate, encourage, equip, and empower those seeking comprehensive natural strategies to heal strong and stay strong.
HealingStrong Connect Groups meet monthly in various areas of the U.S. and South Australia to link others interested in natural strategies, holistic protocols, and local resources. Their groups focus on mind, body and emotional healing based on Biblical promises, as they believe the God of the Bible is our healer. For more information on how to start a group, or become involved in one, please go to: http://www.healingstrong.org/groups, and like us on Facebook at http://www.facebook.com/healingstrong.
Cancer Crackdown is so awesome and helps patients without a lot of money get supplements and treatments and even helps them raise money:
http://cancercrackdown.org/
https://www.facebook.com/CancerCrackdown
Also, the Independent Cancer Research Foundation is another charity that we support, also known as Cancer Tutor. They have been a TREMENDOUS help for cancer patients and have a TON of life saving information they give out freely and they do real research into treatments for cancer that are outside of the toxic Big Pharma Big 3:
http://www.theicrf.com/
The best that we can suggest is to contact Healing Strong and Cancer Crackdown for any need in financial resources.
We also support Mary Beth’s “Nick Gonzalez Foundation” which is bringing Nick’s protocol to the people…which is one of the most powerful cancer killing protocols out there.
I hope this is helpful! Please don’t hesitate to reach out to us again if there’s anything else we can help with.
Wishing you endless blessings and love!
Hi ithank you for the wonderful information that you provide, u have Multiple Myloma for 11/12years been having Chemo the whole time . My question is how long does it take for Keto to start seeing effects, benefits and results once on this diet thank Ty once again 👍
Hi Violet –
We appreciate you reaching out to us about this.
Unfortunately, we are unable to give any kind of medical advice. It would be best if you consult with one of the doctors we interviewed in the Global Quest series and explain your individual case to them.
Please note that we are not able to select an expert for you.
We’ve created a page with the experts’ contact info as it was available to us.
Here’s the link to the actual webpage:
http://thetruthaboutcancer.com/experts-info-sheet/
We do have one more resource you may be interested in. During our Live Event 2017 series, Dr. Patrick Quillin suggested contacting The Institute of Functional Medicine to locate a practitioner in your area.
If you are interested, here is the link to search for a Functional Medicine Practitioner in your area: https://www.ifm.org/find-a-practitioner/
I’m also attaching the info about the Hope4Cancer clinic in Tijuana, MX if you’d like to reach out to them.
http://www.hope4cancer.com/
https://hope4cancer.com/schedule-a-call/
Please also see the resources below in case they will be helpful to you.
From our beginning, we have supported charities that really help these cancer patients.
HealingStrong:
http://healingstrong.com/
https://www.facebook.com/healingstrong
HealingStrong supports MANY cancer patients and are amazing. HealingStrong, a nonprofit organization, is supported 100% by volunteers, who are experienced in using natural strategies. The organization’s mission is to educate, encourage, equip, and empower those seeking comprehensive natural strategies to heal strong and stay strong.
HealingStrong Connect Groups meet monthly in various areas of the U.S. and South Australia to link others interested in natural strategies, holistic protocols, and local resources. Their groups focus on mind, body and emotional healing based on Biblical promises, as they believe the God of the Bible is our healer. For more information on how to start a group, or become involved in one, please go to: http://www.healingstrong.org/groups, and like us on Facebook at http://www.facebook.com/healingstrong.
Cancer Crackdown is so awesome and helps patients without a lot of money get supplements and treatments and even helps them raise money:
http://cancercrackdown.org/
https://www.facebook.com/CancerCrackdown
Also, the Independent Cancer Research Foundation is another charity that we support, also known as Cancer Tutor. They have been a TREMENDOUS help for cancer patients and have a TON of life saving information they give out freely and they do real research into treatments for cancer that are outside of the toxic Big Pharma Big 3:
http://www.theicrf.com/
The best that we can suggest is to contact Healing Strong and Cancer Crackdown for any need in financial resources.
We also support Mary Beth’s “Nick Gonzalez Foundation” which is bringing Nick’s protocol to the people…which is one of the most powerful cancer killing protocols out there.
I hope this is helpful! Please don’t hesitate to reach out to us again if there’s anything else we can help with.
Wishing you endless blessings and love!
Is Keto diet good for anybody, a healthy person?
I’ve been diagnosed with 2 types of Leukemia. CLL Chronic Lymphocytic Leukemia as well as MM Multiple Myeloma. The Keto diet helps me a lot, but do you have a proof that it can cure blood cancer?
Hi Sanet –
Thanks for your comment.
Please note that we don’t claim to cure anything nor do we claim that any diet will be a guaranteed cure to an illness.
Each person’s health and body is different from the other. Therefore it’s always best to consult with a healthcare practitioner so they can diagnose you (or share advice with you) based on your exact body condition.
If you’re interested in learning about whether the Keto diet is good for your body, we would suggest that you consult with one of the doctors we interviewed in the Global Quest series so they can give you the most appropriate advice based on your exact condition.
Please note that we are not able to select an expert for you.
We’ve created a page with the experts’ contact info as it was available to us.
Here’s the link to the actual webpage:
http://thetruthaboutcancer.com/experts-info-sheet/
We do have one more resource you may be interested in. During our Live Event 2017 series, Dr. Patrick Quillin suggested contacting The Institute of Functional Medicine to locate a practitioner in your area.
If you are interested, here is the link to search for a Functional Medicine Practitioner in your area: https://www.ifm.org/find-a-practitioner/
Hope these resources are helpful to you. Blessings and love!
Hi,
Am breast cancer warrior, did chemo and radio therapy and took tamoxifen for 2years. what is the best protocol to prevent recurrence. Peninah, Kenya (Africa)
Hi Peninah –
We appreciate you reaching out to us about this.
And we’re so happy to hear that you’re a cancer warrior – God bless you!
Unfortunately, we are unable to give any kind of medical advice. The best advice we can give you is to consult with one of the doctors we interviewed in the Global Quest series.
Please note that we are not able to select an expert for you.
We’ve created a page with the experts’ contact info as it was available to us.
Here’s the link to the actual webpage:
http://thetruthaboutcancer.com/experts-info-sheet/
We do have one more resource you may be interested in. During our Live Event 2017 series, Dr. Patrick Quillin suggested contacting The Institute of Functional Medicine to locate a practitioner in your area.
If you are interested, here is the link to search for a Functional Medicine Practitioner in your area: https://www.ifm.org/find-a-practitioner/
I’m also attaching the info about the Hope4Cancer clinic in Tijuana, MX if you’d like to reach out to them.
http://www.hope4cancer.com/
https://hope4cancer.com/schedule-a-call/
Along with this, we also have comprised a list of health experts and doctors we’ve interviewed in our Eastern Medicine: Journey to Asia documentary. Ty and the team are still working on collecting the contact information for the rest of these doctors and experts. Please note that this link will be updated with more doctor’s contact info as they share it with us.
https://thetruthaboutcancer.com/eastern-medicine-experts-info-sheet/
Please also see the resources below in case they will be helpful to you.
From our beginning, we have supported charities that really help these cancer patients.
HealingStrong:
http://healingstrong.com/
https://www.facebook.com/healingstrong
HealingStrong supports MANY cancer patients and are amazing. HealingStrong, a nonprofit organization, is supported 100% by volunteers, who are experienced in using natural strategies. The organization’s mission is to educate, encourage, equip, and empower those seeking comprehensive natural strategies to heal strong and stay strong.
HealingStrong Connect Groups meet monthly in various areas of the U.S. and South Australia to link others interested in natural strategies, holistic protocols, and local resources. Their groups focus on mind, body and emotional healing based on Biblical promises, as they believe the God of the Bible is our healer. For more information on how to start a group, or become involved in one, please go to http://www.healingstrong.org/groups, and like us on Facebook at http://www.facebook.com/healingstrong.
Cancer Crackdown is so awesome and helps patients without a lot of money get supplements and treatments and even helps them raise money:
http://cancercrackdown.org/
https://www.facebook.com/CancerCrackdown
Also, the Independent Cancer Research Foundation is another charity that we support, also known as Cancer Tutor. They have been a TREMENDOUS help for cancer patients and have a TON of life-saving information they give out freely and they do real research into treatments for cancer that are outside of the toxic Big Pharma Big 3:
http://www.theicrf.com/
The best that we can suggest is to contact Healing Strong and Cancer Crackdown for any need in financial resources.
We also support Mary Beth’s “Nick Gonzalez Foundation” which is bringing Nick’s protocol to the people…which is one of the most powerful cancer-killing protocols out there.
I hope this is helpful! Please don’t hesitate to reach out to us again if there’s anything else we can help with.
Wishing you endless blessings and love!
Go VEGAN! The longest lived societies all eat largely vegan (fruits, veggies, legumes, grains) with very small amounts of animal products, typically during holidays. They do not eat processed foods. Eat your fruits and veggies!! Based on statistics and scientific research you will live longer eating vegan then animal product heavy diets. Stay away from cholesterol inducing fats, BUTTER AND GRASS FED ANIMALS.
hi ty! obviously, your site is mostly about cancer, but do you have topics on heart disease? or can you recommend one or two?
thanks!
Some cancers utilize fats and glutamine more than glucose. These cancers won’t be helped by a keto diet.
I have lost 98 lbs. in just over a year from 294 to 196. My triglycerides fell from 297 to 110, my A1C which had been at 6.7 for several years fell to 5.5. My short-term memory is still not where I would like it to be but can remember numbers better. My eyesight is better. I walk 5 miles, 3 times per week and earn 27 aerobic points for the duration and distance I walk. Earn another 3 points with other activities. 30 points a week is a good maintenance level for exercise. At one time I could not control my eating and felt like I just didn’t have any man about me, much less integrity or character. Once I found out it was my blood chemistry and insulin resistance from dr. rob Thompson’s book: Insulin Resistance Solution, I started the keto program I’m own since December-29th of last year. My insulin resistance had come down from 47.1 to 19 and hope to get it below 8.